


![]()
Isn’t Japan thought of as an advanced country in the field of medicine?
However, in the current situation it is a fact that much of the medical devices and
pharmaceuticals used in the medical front depend on products from Europe and the U.S.
Under these circumstances, Asahi Intecc is gaining attention from medical professionals
around the world, through its medical devices for catheter treatment and primarily the
guide wires which are the fruits of its original technologies.
We often hear about catheter treatment, but what kind of
treatment is it exactly?
What are the guide wires which are highly valued in the world?
What is the source of the competitiveness of Asahi Intecc which
generates these products?
Here, we provide answers that can be easily understood about
Asahi Intecc’s business, strengths and growth strategy.

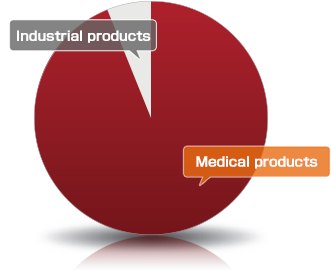
Various wire products are used in the medical device and industrial device fields.
For some people, the word “wire” may not paint a clear picture of these products.
Even so, wire products are used in all sorts of things that surround us, from various medical devices to cars, air conditioners, OA equipment and even in the fields of construction and fishery.
Among these products, Asahi Intecc develops, produces and sells guide wires, guiding catheters and balloon catheters which are medical devices indispensable for catheter treatment.
Some products used in catheter treatment have diameters as thin as 0.35mm.
With its excellent development capabilities and production technologies in the medical device field, Asahi Intecc has a high share not only in Japan but in the world as well.

What exactly is catheter treatment in which Asahi Intecc’s products play active roles?
Catheter treatment is one of the methods of treatment for diseases such as angina pectoris and myocardial infarctions that are caused by the heart’s blood vessels (coronary arteries) becoming clogged or narrowed by cholesterol.
Previously it was common to treat such diseases with medication or, if symptoms were heavy, with coronary artery bypass grafting a major surgery with thoracotomy.
Meanwhile, catheter treatment has been making notable progress in recent years. This is a method of treatment in which narrow tubes called catheters are inserted from the wrist or from the base of the foot to widen the narrowed blood vessels. Since the treatment doesn’t require thoracotomy, there is little pain for patients (minimally invasive) and the economic burden is lighter due to the shortened length of hospital stay which allows early social rehabilitation, and it has thus become the main method of treatment for coronary artery diseases.


Three products used for this treatment are guide wires, guiding catheters and balloon catheters. Asahi Intecc has been contributing to the proliferation of minimally-invasive treatment in recent years through the development, production and sale of these products which are indispensable for catheter treatment.


The market for the field of catheters continues to expand significantly with the spreading of minimally-invasive treatment.
Asahi Intecc’s guide wires, its main product, have now extended to over 110 countries and regions, and the number of cases of catheter treatments are significantly increasing in developing countries centering on BRICs nations as well, due to their economic development.
By accurately adjusting to such changes of the times and supplying products that are indispensable for medical care demanded by the growing market, Asahi Intecc aims for sustainable growth on a global scale.
So, what is the source of the competitiveness of Asahi Intecc which continues to expand its share on a global scale?


The strength of Asahi Intecc is its integrated production system that covers everything from the materials to the finished products. It all began in 1976. At the time, Asahi Intecc was a small factory that produced and sold ultra-fine stainless steel ropes used in industrial equipment in Osaka.
Later, the company entered into the medical device business, a field in which it could use its technologies. A certain offer from a doctor was to be a major turning point towards the big leap forward.
At the time, the majority of treatments for CTO (chronic total occlusion) depended on surgery. Then, a doctor who was a leading expert in catheter treatment asked the company if it could make guide wires that could be used in CTO treatment. Initially the doctor had asked a leading overseas medical device manufacturer, but the task was one which was difficult to accomplish with the technological level at the time. Even among doctors it was not common sense to treat this illness with catheters. Treatment of CTO with catheters was particularly difficult and success would require sophisticated skill on the part of the doctor and a new level of high-functioning guide wires that could accurately express the doctor’s fine senses.

It was an excursion into uncharted territory. However, four core technologies, and particularly a sophisticated torque technology, would be indispensable to be able to faithfully express the fingertip senses of doctors with the guide wires.
With the ultra-fine stainless steel rope technology Asahi Intecc had cultivated, the company’s rapid and detailed experimental and adapting capabilities, in which the voices of doctors on the medical front line were steadily incorporated through trial and error, and returning time and time again to the materials level, as well as with the company’s spirit of craftsmanship dedicated to producing products that could treat this disease, finally in 1995 the company succeeded in developing the first PCI guide wire for CTO treatment in Japan. Following reports by the doctor and others successful treatments at academic societies, as well as its use overseas, Asahi Intecc’s products and technologies started to be known around the world.
Unlike other medical device manufacturers that order components and assemble them, Asahi Intecc has its DNA of an integrated production system that starts from materials and finishes with the final product, as well as a hands-on approach that has been unchanging since its founding. This is what helps realize the speed and experimenting/adapting capabilities unmatched by other companies.



Asahi Intecc’s industrial products, such as its stainless steel ropes for industrial equipment, were in a severe situation right before the company entered into the medical device field, with demands for significant cutting of costs due to the appreciating yen. In order to adjust, Asahi Intecc relocated its production base overseas to Thailand. At the same time, production that added high value, different to what had been before was being required in order for the company to stay alive. Asahi Intecc decided to take on the challenge of launching its medical device business in
order to meet these conditions in the tightness of the times.
As of now, Asahi Intecc has production bases in Thailand, Vietnam and Philippines. By having the base in Japan specialize in R&D and experimentation, while the factories overseas serve the role as bases for mass production, cost competitiveness is strengthened and the injection of large amounts of management resources into R&D is made possible.
By further enhancing R&D that looks far into the future of the medical industry making rapid progress, Asahi Intecc is advancing on the front line of technology as a R&D-type company that can generate excellent products on an ongoing basis.
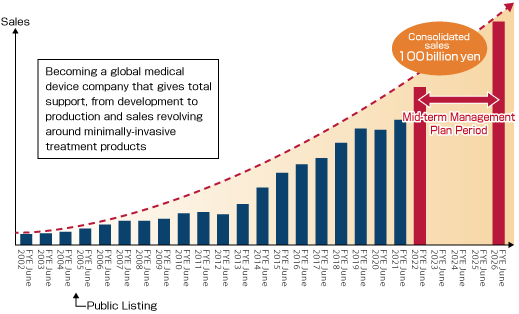
Please click here for details of the New Medium-term Management Plan“ASAHI Going Beyond 1000.”
With the theme of “becoming a global medical device company that gives total support from development to production and sales revolving around minimally-invasive treatment products,” Asahi Intecc is aiming to expand its revenues through enhancing sales capabilities, in addition to producing medical equipment.
We will strengthen our revenue base centering on the medical device field in an aim to become a global medical device company.

![]()
The appearance of innovative PCI guide wires that made CTO treatment possible, was greeted with much surprise from medical professionals around the globe. As the increase of the rate of successful CTO treatments became clear through the case reports of academic associations, all eyes were turning towards Asahi Intecc.
The turning point for the acceleration of overseas development came in 1996 with a request from a doctor, who was a world authority in this field, to sell the products in the U.S. as well.
Even today, active debates and exchanges of heated opinions with doctors are taking place in various medical fronts to be utilized in the development of new products. At the root is the thorough hands-on approach. This is because the needs of doctors cannot be grasped and excellent products would not be able to be generated if the medical fronts are not really understood.
With the results of treatments being announced at academic societies through such doctors and promotional activities for medical technologies being held around the world, the Asahi brand recognition is heightened.


Asahi Intecc’s leading products are currently for the cardiovascular system area, but treatment areas are expanding to encompass other areas such as peripheral vascular (lower-limb), abdominal and cerebrovascular areas.
Some of these products have obtained large shares in Japan and year by year more are being highly valued in the world. We will continue to actively develop overseas through the success cases of new technologies and products in Japan.
Up to now, Europe and the U.S. led the trends in the medical industry. Even in Japan which is considered to be an advanced medical nation, the majority of the medical devices and pharmaceutical products used in the medical front, were developed in the West.
Amidst such trends, Asahi Intecc swept the world with guide wires developed in Japan. With the new market for CTO treatment created through the proliferation of our products, we are aiming to further expand and leap forward.